Introduction
Geneious Prime provides all of the tools required for primer design and in silico simulation of Gateway® cloning.
Thermo Fisher Scientific describe their Gateway® Technology as “a universal cloning method that takes advantage of the site-specific recombination properties of bacteriophage lambda to provide a rapid and highly efficient way to move a gene of interest into multiple vector systems.” See the Thermo Fisher Scientific website for a full description of Gateway® cloning.
To express a gene of interest using Gateway the following steps are taken:
- Generate an entry clone by performing a BP recombination reaction between a pDONR™ vector and an attB PCR product or expression clone.
- Generate the desired expression clone by performing an LR recombination reaction between the entry clone and a Gateway® destination vector.
- Introduce your expression clone into the host system of choice for expression of your gene of interest.
In this tutorial we will learn how to:
- Design Gateway-compatible oligonucleotide primers to amplify a sequence of interest
- Simulate PCR to create a Gateway-compatible PCR product
- Simulate recombination of PCR product with a donor vector via a “BP” reaction to create an Entry vector
- Simulate recombination of an Entry vector with a Destination vector via an “LR” reaction to create an Expression vector
- Simulate multipart Gateway cloning
INSTRUCTIONS
To complete the tutorial yourself with included sequence data, download the tutorial and install it by dragging and dropping the zip file into Geneious Prime. Do not unzip the tutorial.
THE BASICS
Tutorial structure & primer design rules
EXERCISE 1
Designing primers for Gateway cloning
EXERCISE 2
BP Entry clone reaction
EXERCISE 3
LR Destination clone reaction
MULTISITE OVERVIEW
Gateway multisite cloning
EXERCISE 4
Multisite 1: Primer design and PCR
EXERCISE 5
Multisite 2: Entry and Destination vectors
GATEWAY VECTORS
Preparing Gateway vector sequences
The Basics
Tutorial Structure
The first three exercises in this tutorial cover the steps required for simulation of single insert Gateway cloning.
The fourth and fifth exercises cover simulation of multisite Gateway cloning.
The final section describes how to prepare vector sequences for use with the Gateway cloning tool.
Primer Design Rules Overview
Thermo Fisher Scientific recommend that to enable efficient Gateway® cloning, primers for addition of attB sites MUST contain the following structure:
- Four guanine (G) residues at the 5′ end of each primer, followed by a 25 bp attB site, and followed by At least 18-25 bp of template/gene-specific sequence
- The attB1 site should end with a thymidine (T)
- If you plan to express native protein in E. coli or mammalian cells, you may want to include a Shine-Dalgarno or Kozak consensus sequence, respectively, in your PCR primer
- If fusing your sequence in-frame with an N-terminal tag, the forward (attB1) primer must include two additional nucleotides to be in-frame with the attB1 region. The two nucleotides cannot be AA, AG, or GA, as these additions will create a termination codon
- If fusing your sequence in-frame with an C-terminal tag, the reverse primer must include one additional nucleotide to be in-frame with the attB2 region. If you do not want to perform a C-terminal fusion you must include a stop codon
When the Geneious Primer design tool is used to add att extensions to primers it will automatically do the following:
- Add four 5′ guanine (G) nucleotides
- Add the specified att motif to your primers
- End the attB1 with a T “ACAAGTTTGTACAAAAAAGCAGGCT“
- Add a 2 nucleotide “TA” spacer to forward attB1 primers†
- Add a 1 nucleotide “T” spacer to reverse primers†
† In Geneious Prime 2019.1 and earlier “spacer” nucleotides are added as part of the att motif annotation. In future versions of Geneious Prime the “spacer” nucleotides will be added as separate annotations.
The primer design tool will not add translation initiation motif sequences. Users will need to add these motifs manually as extensions during the primer design process.
The primer design tool will not add a stop codon to a reverse primer. If you require a stop codon you should incorporate a template-derived stop codon (if present) or manually add a stop codon as an extension during the primer design process.
Exercise 1: Designing Primers for Gateway Cloning
In this exercise we will design oligonucleotide primers to amplify the mature xynB CDS.
The forward and reverse primers will be designed to incorporate attB1 and attB2 sites respectively, to allow clonase-mediated integration of the PCR product into a Gateway entry vector. The primers will be designed to both have a first-round melting temperature (Tm) of 55°C.
The primers will be designed to precisely amplify the mature xynB CDS (no signal sequence) to ensure that when the xynB CDS is transferred into the pDest17 destination vector it will be positioned for in-frame fusion to the vector start codon and associated HIS-tag sequence. This will ensure optimal transcription and expression of the XynB gene product. The xynB CDS stop codon will be included to ensure correct termination of the gene product.
Exercise
1. To start the exercise select the DTU76545 sequence to view it.
2. Click on the yellow xynB mature CDS annotation to select the mature CDS region (positions 139 to 1149).
(If the xynB mature CDS annotation is not visible then click on the ‘yellow arrow’ icon in the Sequence View side panel and check the option to Show Annotations.)
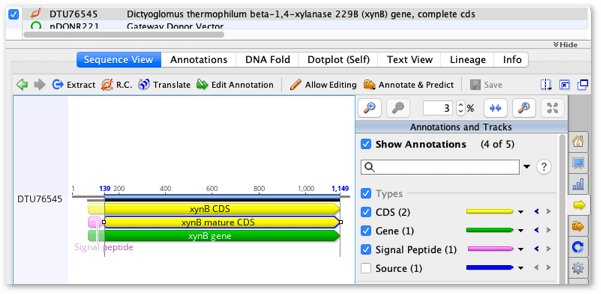
3. With the xynB mature CDS annotation selected, click the Primers button on the Toolbar and choose Design new primers.
4. In the Design New Primers setup window:
- Click on the cog icon in the bottom left corner of the Window and choose to Reset to Defaults
- Ensure Select Task: is set to Design New
- Ensure the Forward Primer and Reverse Primer options are checked
- Set Task: to Precise
- Ensure the Included Region: option is checked and the range is set to 139 to 1149
- Set Number of pairs to generate to 1
We will design primers with a melting temperature of around 55°C so we need to changes Tm Min: default settings:
- set the Tm Min: / Optimal: / Max: values to 50 / 55 / 60
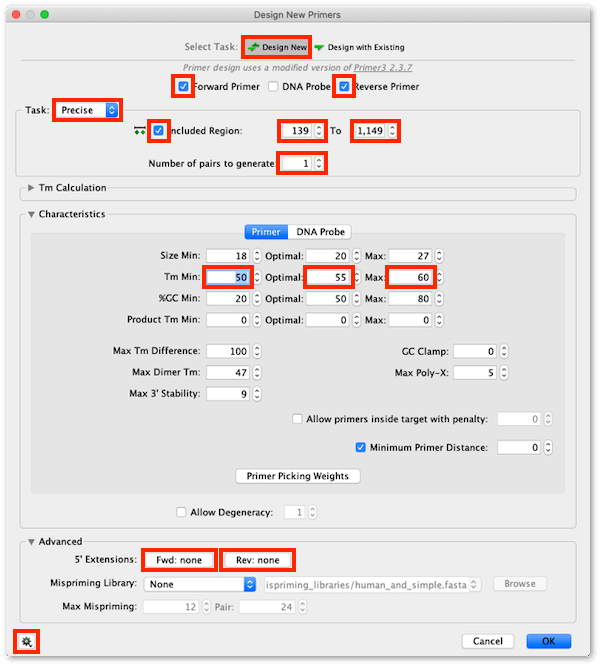
5. To add Gateway extensions to the primers, expand the Advanced section of the Setup window, then click on the 5′ Extensions: Fwd: button, click the Gateway site… button and from the drop down menu choose Site: attB1.
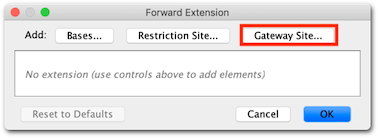
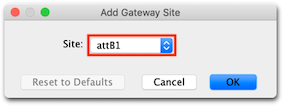
Click OK, and you will see that the extension will comprise 4 G nucleotides and an attB1 site. The G nucleotides are added to ensure the clonase enzyme can efficiently bind to the terminal aatB1 site.
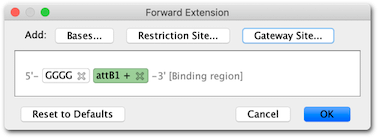
6. For the reverse primer, click the Rev: button, click the Gateway site… button and from the drop down menu choose to add an “antisense” attB2 site.
Click to run the Primer design tool and two new annotations, 139 F and 1149 R will be added to the sequence. Hit Save (Command or CTRL-S) to save the new annotations.
Hover over each primer sequence and you will see a yellow tool tip containing information about the primer.
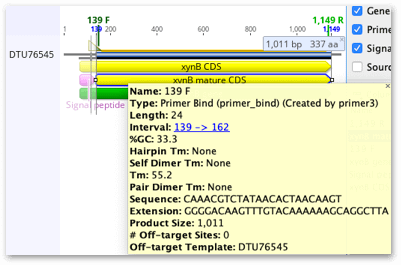
7. The next step is to generate a PCR product sequence. Select the DTU76545 file, click on the Primers button and choose Extract PCR Product. The tool will detect the new forward and reverse primer annotations on the sequence and select them. In the dialog that opens, confirm the Forward and Reverse primers are correctly selected, then hit OK.
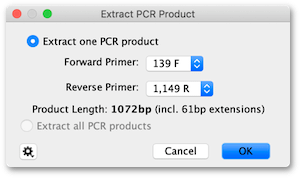
A new DTU76545 PCR Product sequence will be created. Select it and you should see it has inward facing attB1 and attB2 sites positioned and oriented correctly for a BP clonase reaction.
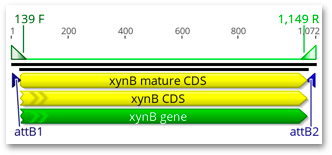
We will use this PCR product in the next Exercise.
If you plan to routinely design Gateway Primers then you can save your ideal primer design settings by clicking on the cog icon in the bottom left corner of the Design New Primers window and choose to Save current settings. You can load these settings next time by clicking on the cog icon and choosing Load Profile.
To extract your primers to individual files select each primer_bind annotation and Hit the Extract button. You can use these extracted sequences for ordering from your oligo synthesis service provider.
Exercise 2: Simulating a “BP” Entry clone reaction
In this exercise we will simulate a BP clonase reaction between our new PCR product and a donor vector with appropriate attP1 and attP2 sites.
Select the pDONR221 sequence to view it. You will see this vector has attP1 and attP2 sites flanking a chloramphenicol (cmR) resistance gene and a ccdB toxin gene. This region will be replaced by recombination with the PCR product to create an entry vector.
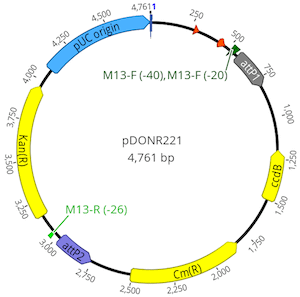
To perform the BP clonase reaction, select the pDONR221 and DTU76545 PCR Product sequences then go Cloning → Gateway Cloning. The Gateway cloning tool will identify the att sites present on both the vector and the PCR product insert, confirm they are appropriately oriented, and inform that a BP reaction will be performed.
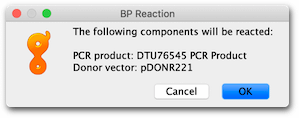
Hit OK and a new sequence called pDONR221 – DTU76545 PCR Product Entry clone will be created. This new 3558 bp sequence represents the expected entry plasmid generated by the clonase reaction. You will see that the clonase reaction has created flanking attL1 and attL2 sites suitable for use in a clonase-mediated LR-reaction.
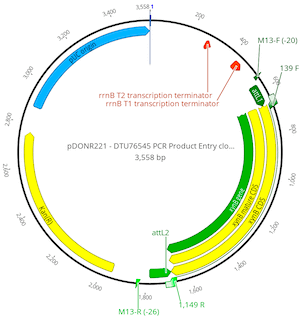
The pDONR vectors manual recommends that individual E. coli colonies containing your clonase-derived constructs are checked by PCR using the M13-F (-20) and M13-R primers. Binding sites for these primers are present and annotated onto the pDONR221 vector. We will now use the Extract PCR Product tool to simulate this PCR reaction so that we can determine the size and sequence of the predicted product.
Select the new pDONR221 – DTU76545 PCR Product Entry clone, Hit the Primers button and choose Extract PCR product. Set the Forward primer to M13-F (-20) and the Reverse primer to M13-R (-26). This will create a 1303 bp PCR product sequence.
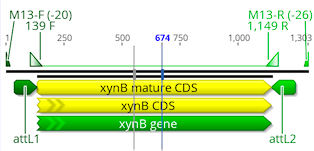
Exercise 3: Simulating a “LR” Destination clone reaction
In this exercise we will simulate a LR clonase reaction between our new pDONR221 – DTU76545 PCR Product Entry clone and the destination vector pDEST17 vector.
Select the pDEST17 sequence to view it. You will see this vector has attR1 and attR2 sites flanking a chloramphenicol (cmR) resistance gene and a ccdB toxin gene.
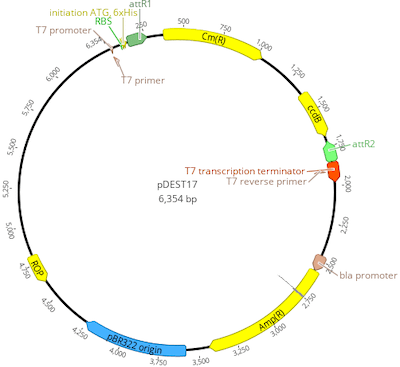
To perform the BP clonase reaction, select the pDEST17 and pDONR221 – DTU76545 PCR Product Entry clone sequences then go Cloning → Gateway Cloning. The Gateway cloning tool will identify the att sites present on the entry vector and Destination vector and confirm an LR reaction can be performed. In the test tube an LR reaction creates two new plasmid species. The Gateway tool will output both plasmids if you wish. The tool will ask if you want to “keep both products of the reaction”. In this exercise leave this option unchecked so only the final Expression vector will be created.
The tool will confirm sites in each vector are appropriately oriented, and inform that a LR reaction will be performed.
Hit OK and a new 5713 bp plasmid called pDEST17 – pDONR221 – DTU76545 PCR Product Entry clone Expression clone will be created.
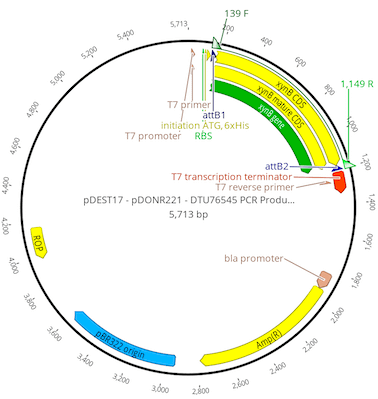
The new file name, although descriptive is a bit long and cumbersome. Select the file in the document table, click on the file name to edit it and rename the file to pDEST17:xynB mature.
As a last step we will check that our insert is in-frame with the vector start codon and HIS-tag.
Select the renamed pDEST17:xynB mature sequence, ensure Translation is turned on in the Display tab, then zoom in so that you can see the initiation ATG annotation at the 5′ end of the xynB mature CDS annotation. Click on the xynB mature CDS annotation to select it, then drag the left hand boundary of the annotation to extend it to line up with the first “A” of the start codon. You will see in the translation that the start codon and HIS-tag are in-frame with the translated start of the xynB mature CDS “QTSI…”.

Note that changing annotations on a sequence with actively linked parents will break the linkage. When you adjust the xynB mature CDS annotation boundary you will get a warning about Actively linked parents. You can ignore this warning and click Continue Editing, just remember to not save the document when asked, or alternately, use menu File → Save As to save an unlinked copy of the original document.
Performing the BP and LR reactions in a single step
To simplify your Gateway reactions it is possible to use the Gateway cloning tool to perform the BP and LR reactions in a single operation. Simply select your PCR product insert with attB sites, your pDONR vector and your pDest vector and go Cloning → Gateway Cloning.
To try cloning in one step, select the DTU76545 PCR Product, the pDONR221 vector and the pDEST17 vector and go Cloning → Gateway Cloning.
The Gateway cloning tool will confirm the selected sequences can be “reacted” and will ouput the same final 5713 bp expression plasmid.
Overview: Simulating Gateway multisite cloning
Thermo Fisher Scientific provide a number of specialised pDONR vectors that allow multiple “parts” (2, 3 or 4) to be recombined, in a specific order and orientation, with a destination vector. The Geneious Prime Gateway Cloning tool allows simulation of multisite Gateway reactions.
See the MultiSite Gateway® Pro manual for detailed information on multisite Gateway cloning.
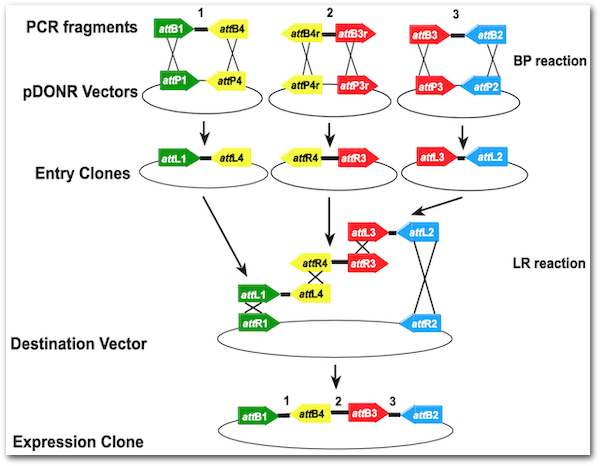
The steps required for multisite cloning parallel those performed in Exercises 1, 2 and 3:
- Design primers for PCR amplification and addition of unique attB sites to each part.
- Create individual entry clones for each part using specific multisite pDONR vectors.
- Recombine the 3 entry clones with a destination vector using the Gateway cloning tool.
The Gateway clonase recognition sites are directional and must be orientated appropriately in order for multisite recombination to proceed correctly. If you examine the above figure you will see that in a 3 part clonase reaction the PCR products for parts 1 & 3 have attB sites oriented to point inwards, whereas for part 2 the attB sites are oriented to point outwards.
For multisite gateway cloning, variants of the attB recognition site are used for each part, in the case of a 3 part reaction: attB1+attB4, attB4R+attB3R and attB3+attB2.
The following table is provided to assist when using the Geneious primer design tool to ensure you select the correct att sites when adding to primers for amplification of multisite Gateway parts.
| Vector and recombination type | left/right att sites | att site to add to forward primer | Att site to add to reverse primer |
|---|---|---|---|
| pDONR221 P1-P5R | |||
| 2-fragment – part 1 | attB1 | Sense: attB1 | |
| 4-fragment – part 1 | attB5r | Antisense: attB5r | |
| pDONR2221 P5-P2 | |||
| 2-fragment – part 2 | attB5 | Sense: attB5 | |
| attB2 | Antisense: attB2 | ||
| pDONR221 P1-P4 | |||
| 3-fragment – part 1 | attB1 | Sense: attB1 | |
| attb4 | Antisense: attB4 | ||
| pDONR221P4R-P3R | |||
| 3-fragment – part 2 | attB4r | Sense: attB4r | |
| 4-fragment – part 2 | attB3r | Antisense: attB3r | |
| pDONR221 P3-P2 | |||
| 3-fragment – part 3 | attB3 | Sense: attB3 | |
| 4-fragment – part 3 | attB2 | Antisense: attB2 | |
| pDONR221 P5-P4 | |||
| 4-fragment – part 4 | attB5 | Sense: attB5 | |
| attB4 | Antisense: attB4 |
Exercise 4: Multisite Part 1: Primer design and PCR
The files for this exercise are provided in a subfolder called Multisite Gateway cloning. Select this folder to see the example files required for this exercise in the Document Table.
As part of the work described by Peterson and Stowers (2011), a Gateway multisite reaction was used to place the yeast GAL4 CDS between two regulatory elements derived from the D. melanogaster trpA1 gene. The final Gateway construct was then integrated into into the D. melanogaster genome to demonstrate that the trpA1 regulatory elements upregulated GAL4 expression, and consequently UAS-GFP (green fluorescent protein) expression, in larval sensory neurons.
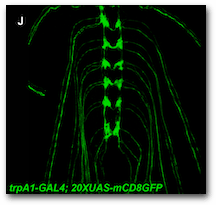
In this exercise we will simulate the work of Peterson and Stower and use the Geneious Gateway tool to perform a Gateway multisite reaction to combine separate upstream and downstream D. melanogaster trpA1 regulatory elements (trpA1 UP and trpA1 Down) with the yeast GAL4 transcriptional activator CDS and with the D. melanogaster-specific destination vector pDESTHaw.
The required part order (with att sites used) is depicted below:
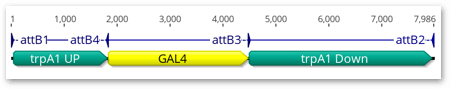
In this exercise we will:
- Design primers for PCR-mediated addition of unique attB sites to each of the trpA1 UPstream, GAL4, and trpA1 DOWNstream parts. Primer binding regions will be designed to have a predicted Tm of around 55°C.
- Create individual entry clones for each part using specific multisite pDONR vectors.
- Recombine the 3 entry clones with the destination vector pDESTHaw, using the Gateway cloning tool, to create a sequence suitable for PhiC31 integrase (attB)-mediated integration into the genomes of D. melanogaster embryos.
Part 1: Primer design and PCR
Step 1: Primer design for trpA1 5′ regulatory element (trpA1 UP)
The trpA1 5′ regulatory element will be the first part of a 3 part reaction. As per the table in the Multipart Overview we need to use PCR to add flanking attB1 and attB4 sites to the first trpA1 UP sequence.
The 5′-terminus of the trpA1 UP sequence contains a short palindromic motif that results in primers designed across this region having a high predicted Hairpin Tm. To successfully design PCR primers to amplify this sequence using the Design new Primers tool we will need to adjust the default Primer Design settings.
Select the trpA1 UP sequence, click the Primers button in the Toolbar and choose Design new Primers.
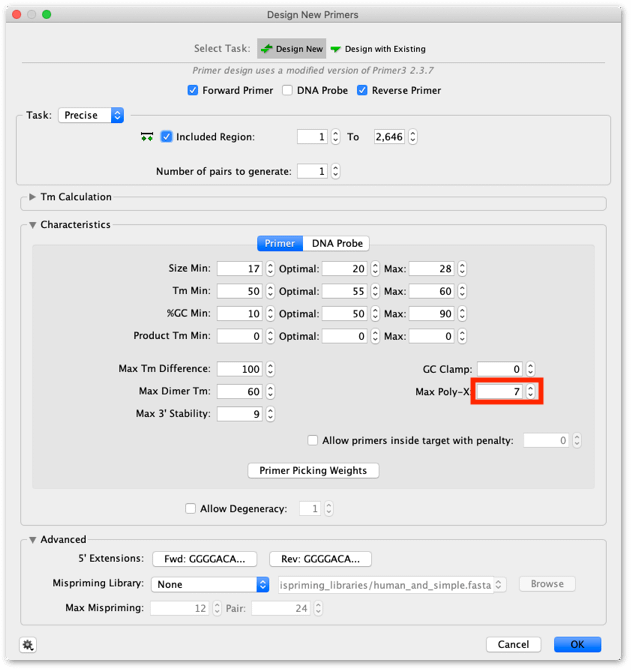
Use the settings shown below. Make sure the Max dimer Tm setting is high (60°C) to ensure the forward primer design does not fail due to the presence of the palindromic motif.
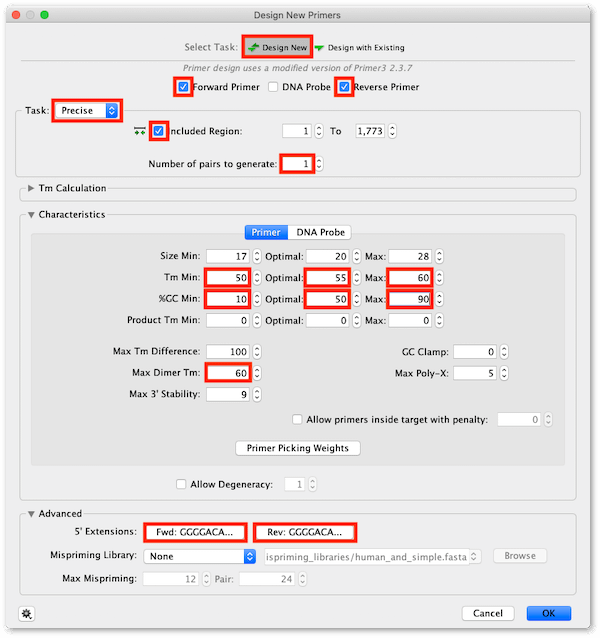
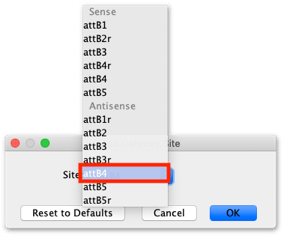
To add Gateway extensions to the primers, expand the Advanced section of the Setup window, then click on the 5′ Extensions: Fwd: button, click the Reset defaults to remove any previous settings, then click the Gateway site… button and from the drop down menu choose Site: attB1.


Click OK, and you will see that the extension will comprise 4 G nucleotides and an attB1 site.

Click the 5′ Extensions Rev: button, click the Reset to Defaults button to clear existing settings, then click the Gateway site button and add an antisense AttB4 site.
Click OK and OK again to run the Primer Design tool and new forward and reverse primers should be added to the sequence. Hit Save.
Now select the trpA1 UP fragment and go Primers -> Extract PCR Product to generate the PCR product amplified by the new primers. This PCR product will be used in the next section for recombination with the entry vector pDONR221 P1-P4.
Step 2: Primer design for GAL4 CDS sequence
The GAL4 CDS will be the second part of the 3 part multisite reaction. As per the table in the Multipart Overview we need to add flanking attb4r and attb3r sites to the GAL4 sequence.
Select the GAL4 sequence file, and click the Primers button in the Toolbar and choose Design new Primers.
Perform the same steps as in Step 1, this time adding a sense attB4r site to the forward primer and an antisense attB3r to the reverse primer.
The GAL4 sequence contains a 7 nucleotide polyA-region at it’s 3′ end in the region where a primer will be designed. To prevent the Primer Design tool failing due to the poly-A region set the Max Poly-X value to 7.
Once primers are added to the GAL4 sequence, select and go Primers -> Extract PCR Product to generate the PCR product amplified by the new primers. This PCR product will be used in the next section for recombination with the entry vector pDONR221 P4r-P3r.
Step 3: Primer design for trpA1 3′ regulatory element (trpA1 DOWN)
The trpA1 DOWN 3′ regulatory region will be the third part of a 3 part multisite reaction. As per the table in the Multipart Overview we need to add flanking attB3 and attB2 sites to the trpA1 DOWN sequence.
Select the trpA1 DOWN sequence file, and click the Primers button in the Toolbar and choose Design new Primers.
Perform the same steps as those in step 1, this time adding a Sense attB3 site to the forward primer and an antisense attB2 to the reverse primer.
Once primers are added to the trpA1 DOWN sequence, select and go Primers -> Extract PCR Product to generate the PCR product amplified by the new primers. This PCR product will be used in the next section for recombination with the entry vector pDONR221 P3-P2.
You should now have three PCR products ready for integration into donor vectors.
Exercise 5: Multisite Part 2: Creation of Entry and Destination vectors
Part 2: Creation of Entry vectors
We will now combine our new PCR products with the appropriate donor vectors using the Gateway Cloning tool.
Select the new trpA1 UP PCR product and the pDONR221 P1-P4 vector and go Cloning → Gateway Cloning. This will create a new circular vector called pDONR221 P1-P4 – trpA1 UP PCR Product Entry clone.
Select the new GAL4 PCR product and the pDONR221 P4r-P3r vector and go Cloning → Gateway Cloning tool. This will create a new circular vector called pDONR221 P4r-P3r – GAL4 PCR Product Entry clone.
Select the new trpA1 DOWN PCR product and the pDONR221 P3-P2 vector and go Cloning → Gateway Cloning tool. This will create a new circular vector called pDONR221 P4r-P3r – GAL4 PCR Product Entry clone.
We now have 3 pDONR entry vectors that can be recombined with an appropriate destination vector in a multisite reaction.
Part 3: Creation of the final destination vector
To create the final destination vector, select pDESTHaw vector and the three new entry clone sequences. Go Cloning → Gateway Cloning. If all vectors have compatible attB sites then you will see the following dialog:
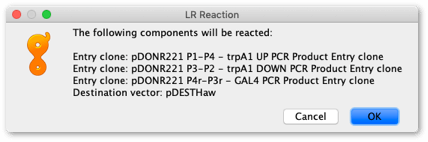
Click OK and a new 15,894 bp circular sequence will be created called pDESTHaw – Reaction of 3 fragments Expression clone.
Rename the sequence to something more useful, select the pDESTHaw – Reaction of 3 fragments Expression clone.
Performing the multisite BP and LR reactions in a single step
To simplify your Gateway multisite reactions it is possible to use the Gateway cloning tool to perform all BP and LR reactions in a single operation. Simply select your three PCR products generated earlier with attB sites, the corresponding 3 pDONR vectors and the pDestHaw destination vector and go Cloning → Gateway Cloning.
The Gateway cloning tool will confirm the selected sequences can be “reacted” and create the same 15,894 bp pDESTHaw-based expression plasmid.
Gateway Vectors: Preparing Gateway vector sequences
The Thermo Fisher Scientific website provides the sequences for their Gateway vectors. However they are not available in a useful annotated form.
If you obtain and import an unannotated Gateway vector sequence then you can use the Geneious Annotate from: tool to identify and annotate various features onto the sequence. For more information our tutorial on Transferring annotations which can be downloaded from https://www.geneious.com/tutorials.
The Geneious Gateway cloning tool requires that Gateway vectors and inserts are annotated with special att annotations. For example, an attR1 motif should be annotated with an annotation of Type: AttR1 and Name: attR1.
To add correct att annotations to your vector sequences, select your new vector sequence and go Cloning→Annotate att sites.
If you receive an annotated Gateway sequence that appears to have att annotations, but it is not accepted by the Gateway cloning tool then double check each att annotation and ensure the correct Type is set.
If you are unsure if the att annotations are correct then select and delete them then use the Cloning→Annotate att sites operation to identify and add the appropriate att site annotations to your sequence.